
How to get an Indian copy of birth certificate from abroad?
Ever wondered how to get your Indian birth certificate while sitting miles away in another country? Well, it's not as challenging as trying to cook the perfect Biryani! First, you need to visit the official website of the Indian consulate in your residing country, which is easier than finding a parking spot in New York city on a Monday! Next, fill up the required online form, attach the necessary documents, and voila, the process is started. The Indian government will then verify your details quicker than your mom can find out about your secret weekend plans! So, sit back and relax, your birth certificate will soon be on its way to you.
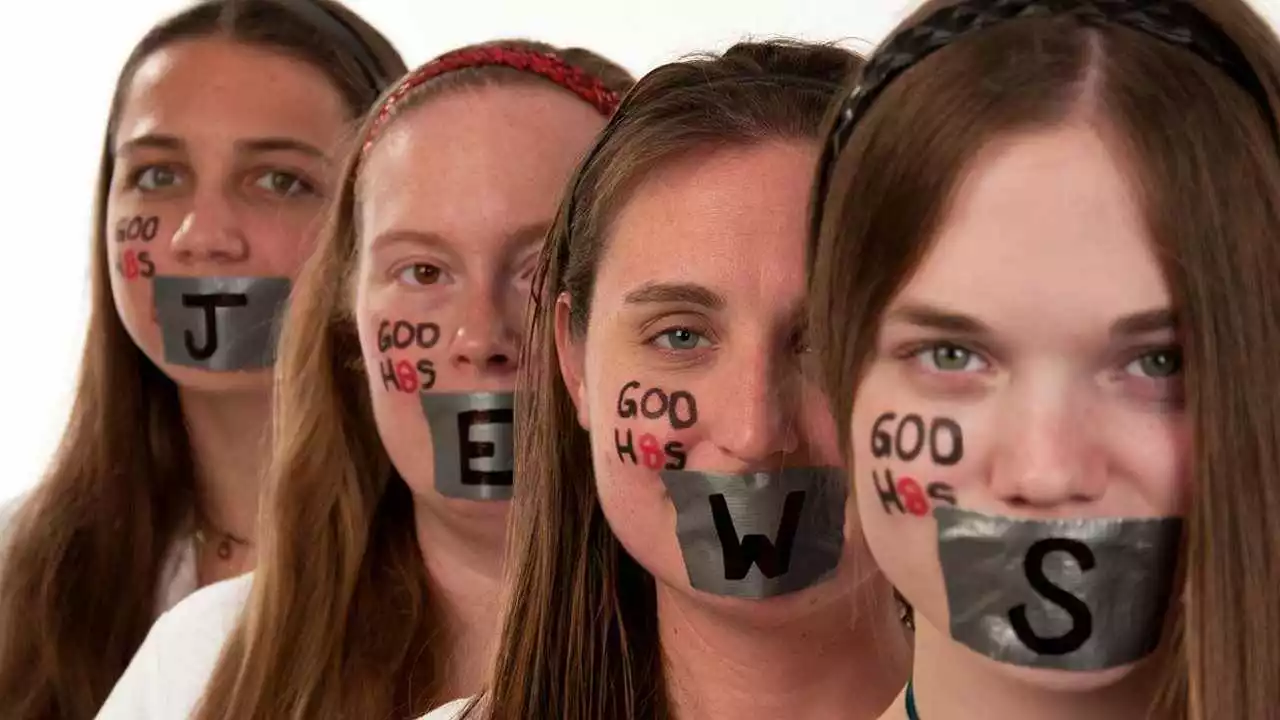
Why do Indians hate themselves and their country so much?
In this blog post, we delve into the complex issue of self-loathing and dissatisfaction among some Indians regarding their own country. While it's important to note not all Indians feel this way, certain factors such as corruption, poverty, and social injustice are often cited as reasons for discontent. Some individuals express dissatisfaction with the country's slow pace of development and the perceived lack of opportunities. Other factors include the societal pressures and the struggle to meet high expectations. It's a multifaceted issue that requires understanding and empathy, not only from fellow Indians but from the global community as well.

Which is India's best online life coach website?
After scouring the internet for the best online life coach platforms in India, I've come to realize that BetterLYF.com is a frontrunner. This platform offers comprehensive life coaching services, ranging from career guidance to relationship advice, all facilitated by experienced professionals. It's easy to use, has flexible time slots, and offers both voice and video call options for sessions. Their unique approach to addressing individual needs sets them apart. In my opinion, if you're in India and looking for some guidance and support, BetterLYF.com is a site you definitely need to check out.

Supreme Court to hear plea seeking President's Rule in Bengal?
In an interesting turn of events, the Supreme Court will be hearing a plea for implementing President's Rule in Bengal. This plea comes amidst a backdrop of political turmoil and heated tension in the area. The petitioner is advocating for the central government's intervention, citing a breakdown of constitutional machinery in the state. It's a significant development that could have far-reaching implications on the state's political scenario. We'll be keeping a close eye on this unfolding story.

How difficult is it to be a vegan in India?
Being a vegan in India can be quite challenging, as the majority of traditional dishes rely heavily on dairy products. However, it is not impossible, as there are numerous plant-based alternatives available in the market. The key is to educate oneself and find resources that cater to vegan diets. Additionally, there is a growing number of vegan-friendly restaurants and communities to connect with. Overall, while it might take some time and effort to adapt, I believe it is becoming increasingly easier to be a vegan in India.

Is it better to work in US or in India?
As a blogger, I have explored the topic of whether it's better to work in the US or India. Both countries have their own unique advantages and drawbacks. The US is known for its higher salaries, better infrastructure, and career growth opportunities, whereas India offers a lower cost of living, strong family ties, and a rich cultural experience. Ultimately, the decision to work in either the US or India depends on an individual's personal goals and priorities. It's important to consider factors such as financial stability, work-life balance, and cultural preferences before making a decision.
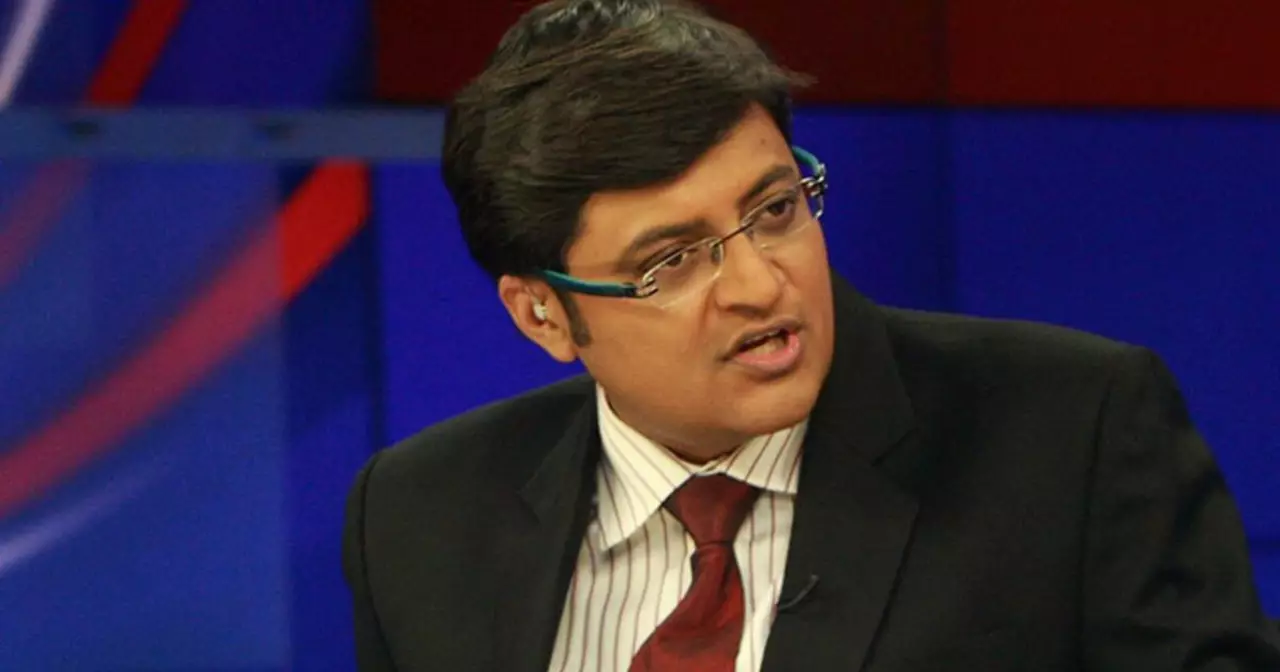
Who is India's most controversial news anchor?
India’s most controversial news anchor is Arnab Goswami. He is renowned for his no-holds-barred approach to journalism, which often leads to heated debates on his show. He has been both praised and criticized for his aggressive style of questioning and his relentless pursuit of the truth. He often pushes boundaries and is not afraid to challenge powerful figures. His willingness to speak out against social injustice and corruption has made him a beloved figure among many Indians. His show has become a staple of Indian news, and his unique style of journalism has made him one of the most influential figures in Indian media.

The Hindu, The Times of India or The Hindustan Times?
The Hindu, The Times of India and The Hindustan Times are three of the most prominent and widely-read English-language newspapers in India. The Hindu, established in 1878, is renowned for its in-depth and unbiased coverage of news, politics, economy and culture. The Times of India, founded in 1838, is the world's largest circulated English-language newspaper and is known for its comprehensive coverage of national and international news. The Hindustan Times, launched in 1924, is one of the most widely read and highly respected newspapers in the country and offers a wide variety of news and opinion pieces. All three papers provide readers with independent, thought-provoking content and are a great source of information and news for Indian citizens.

What are some easy Indian recipes that everyone can cook?
This article provides a selection of easy-to-follow Indian recipes that everyone can make. It outlines a variety of dishes such as Dal Makhani, Aloo Gobi, Samosa, Paneer Kofta Curry and Vegetable Biryani. It also provides step-by-step instructions, ingredient lists and tips to ensure the dish turns out perfectly. With these simple recipes, anyone can enjoy the flavors and aromas of India from the comfort of their own home.
